“ On the Constitution of Atoms and Molecules.” Philosophical Magazine Series 6 26 (July 1913): 1-15.Ĭathcart, B. On the Constitution of Atoms and Molecules. Niels Bohr: His Life and Work as Seen by His Friends and Colleagues. Suspended in Language: Niels Bohr’s Life, Discoveries, and the Century He Shaped. 6.1, “Waves and Electromagnetic Energy.” Electrons change orbits by radiating or absorbing photons.Electrons in stable orbits do not radiate.Orbital angular momentum is quantized hence only certain orbits are possible.Electrons follow circular orbits around a nucleus.Explanation of blackbody radiation and atomic spectra.Majority of the mass is found in the nucleus.Conclusions from the gold foil experiment.Electrons are distributed uniformly throughout the atom.He defines the different isotopes of hydrogen. He details Bohr’s postulates for the hydrogen atom and discusses how the Planck-Einstein relationship applies to electron transitions. 
Sadoway talks about the principles of modern chemistry and how that led to the understanding of the structure of the atom. Periodic Table and Table of Constants Lecture Summary Line spectra the Bohr model uses of emission and absorption spectra 6.3, “Atomic Spectra and Models of the Atom.” 6.2, “The Quantization of Energy.”īlackbody radiation the photoelectric effect The electron radioactivity the atomic model Understand Bohr’s quantization condition.Īrchived Lecture Notes #1 (PDF), Sections 1-3.Understand Rutherford’s “nuclear” model.Understand Thomson’s “plum pudding” model.Learning ObjectivesĪfter completing this session, you should be able to: Sadoway discusses the atomic spectra of hydrogen ( Session 4). Lanthanum (La), magnesium (Mg), chlorine (Cl), titanium (Ti), helium (He), hydrogen (H)īefore starting this session, you should be familiar with: Thomson, proton, electrical charge, amber, alpha particle, beta particle, ionization, conservation of mass, Johannes Geiger, Ernest Marsden, coulomb, Niels Bohr, Bohr model of hydrogen, energy quantization, orbital angular momentum, Planck-Einstein relationship, joule, Newtonian force, Coulombic force, Max Planck, photon, energy, frequency, Planck’s constant, isotope, Henry Cavendish, Harold Urey, Ernest Rutherford, blackbody radiation Lanthanides, actinides, electron, mass, J. Thomson’s plum pudding model, Rutherford’s model of the nucleus, Bohr’s model of the hydrogen atom, Rutherford-Geiger-Marsden experiment, Planck-Einstein relationship, isotopes of hydrogen Thus, the columns of the periodic table represent the potential shared state of these elements' outer electron shells that is responsible for their similar chemical characteristics.Structure of the Atom 3. When an atom gains an electron to become a negatively-charged ion this is indicated by a minus sign after the element symbol for example, \(F^-\). Group 17 elements, including fluorine and chlorine, have seven electrons in their outermost shells they tend to fill this shell by gaining an electron from other atoms, making them negatively-charged ions. When an atom loses an electron to become a positively-charged ion, this is indicated by a plus sign after the element symbol for example, Na +. As a result of losing a negatively-charged electron, they become positively-charged ions. 
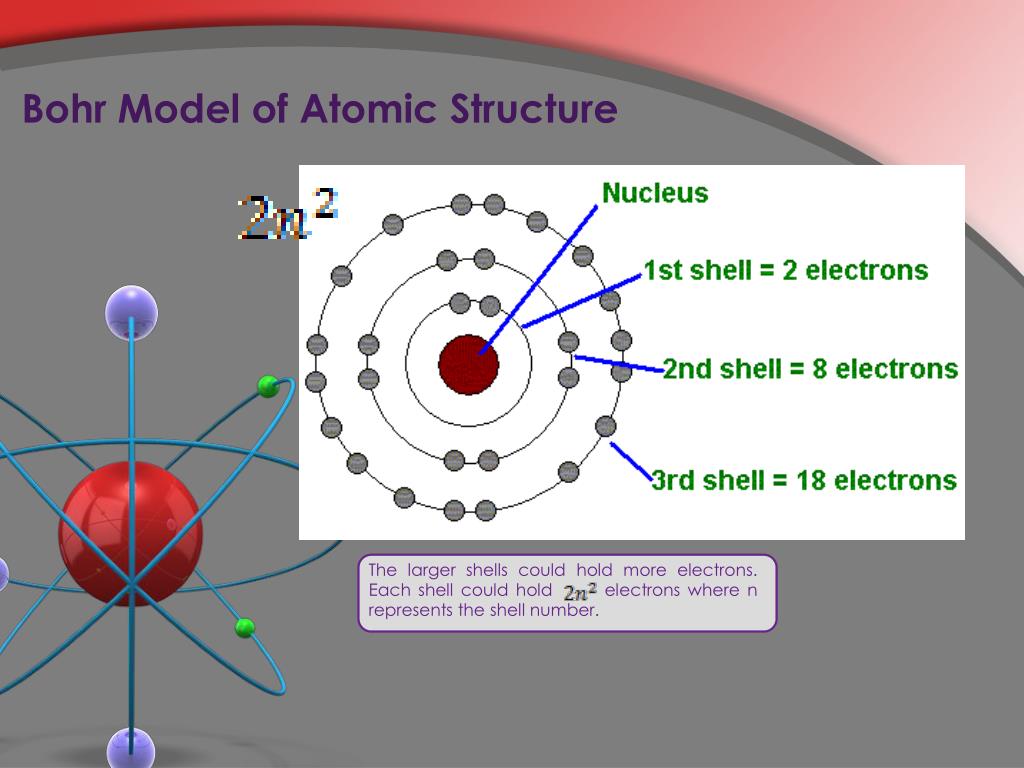
This means that they can achieve a stable configuration and a filled outer shell by donating or losing an electron. In comparison, the group 1 elements, including hydrogen (H), lithium (Li), and sodium (Na), all have one electron in their outermost shells. Their non-reactivity has resulted in their being named the inert gases (or noble gases). As shown in, the group 18 atoms helium (He), neon (Ne), and argon (Ar) all have filled outer electron shells, making it unnecessary for them to gain or lose electrons to attain stability they are highly stable as single atoms. The periodic table is arranged in columns and rows based on the number of electrons and where these electrons are located, providing a tool to understand how electrons are distributed in the outer shell of an atom. Elements in other groups have partially-filled valence shells and gain or lose electrons to achieve a stable electron configuration.Īn atom may gain or lose electrons to achieve a full valence shell, the most stable electron configuration. A full valence shell is the most stable electron configuration. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. \):īohr diagrams indicate how many electrons fill each principal shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
